Hydrogen Bonds in Water: The life was originated and started its evolution in water. Without water, life could not have existed on this planet. The properties of water, both physical and chemical, enabled water as the ‘solvent of life’. The water possesses some unusual physical and chemical properties. These ‘unusual properties’ of water makes water as the solvent of life. The unusual properties of water are due to presence of Hydrogen Bonds in them.
The present post describes the method of formation of hydrogen bonds in water its properties.
How Hydrogen Bond is formed in Water?
Ø Water is a polar solvent.
Ø The polarity of a molecule due to uneven of distribution charges in them.
Ø Uneven charge distribution causes a dipole formation.
Ø One part (pole) of water molecule is slightly positive.
Ø The other part (pole) of water molecule is slightly negative.
Ø This type of difference in the distribution of positive and negative charges in a molecule is due to the huge difference in the electronegativity of the atoms in them.
Ø Electronegativity is the ability of an atom to attract bonded pair of electrons towards its nucleus.
| You may also like NOTES in... | ||
|---|---|---|
| BOTANY | BIOCHEMISTRY | MOL. BIOLOGY |
| ZOOLOGY | MICROBIOLOGY | BIOSTATISTICS |
| ECOLOGY | IMMUNOLOGY | BIOTECHNOLOGY |
| GENETICS | EMBRYOLOGY | PHYSIOLOGY |
| EVOLUTION | BIOPHYSICS | BIOINFORMATICS |
Ø Chemically water is a molecule of hydrogen and oxygen (H2O).
Ø Two hydrogen atoms are covalently bonded to an oxygen atom
Ø Oxygen is a highly electronegative atom when compared to hydrogen.
Hydrogen Bonds in Water
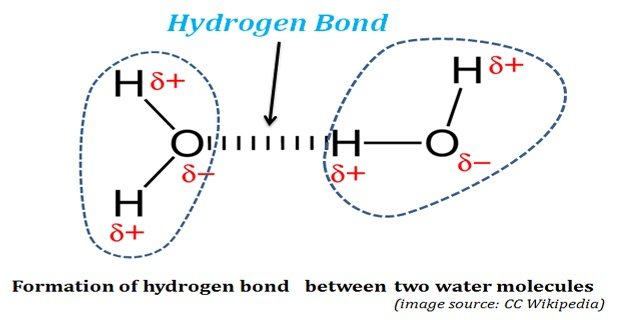
Ø Due to the high electronegativity of oxygen, the oxygen will pull the electron of bonded hydrogen towards its nucleus.
Ø Since electrons are negatively charged, the attracted electron cloud gives a partial negative charge on the side of oxygen represented as δ – ive.
Ø This shift of electrons towards the oxygen also causes a partial positive charge on the side of hydrogen and it is represented as δ+ ive.
Ø Since in a water molecule, the oxygen is covalently bonded to two hydrogen atoms there will be two δ-ive charges in the oxygen and two δ+ive charge in each of the hydrogen.
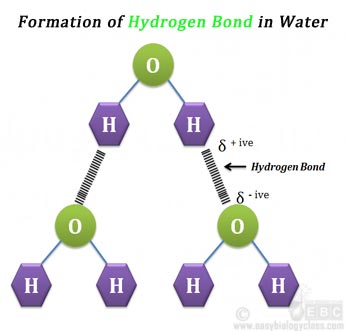 Ø This creates opposite charges in the two poles of the water molecule (dipole).
Ø This creates opposite charges in the two poles of the water molecule (dipole).
Ø Due to the dipole formation, the water molecule can have weak attraction for each other.
Ø When opposite charges are coming close, they can attract each other.
Ø Thus individual water molecules can attract and ‘stick’ like magnets.
Ø This electrostatic attraction in the water molecule is called HYDROGEN BOND.
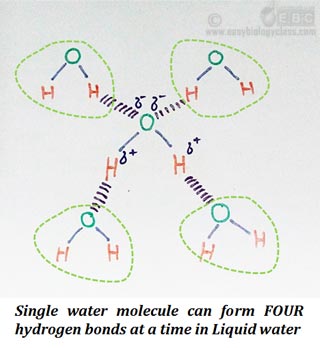
Ø A single water molecule can form FOUR Hydrogen bonds with four different water molecules.
What is hydrogen bond?
Ø It is a weak electrostatic attraction between a hydrogen and an electro-negative atom.
Ø Hydrogen bond is an electrostatic attraction between a hydrogen atom, which is covalently bound to a high electronegative atom (such as Oxygen and Nitrogen), to another electronegative atom of same or different molecules of their close vicinity.
What are the properties of hydrogen bond?
Ø Hydrogen bond is about 10% covalent, due to overlaps in the bonding orbitals, and about 90% electrostatic.
Ø Hydrogen bond is a very weak bond.
Ø It is much weaker than the covalent bond and ionic bond.
Ø Hydrogen bond formed within a molecule is called intramolecular hydrogen bond.
 Ø Hydrogen bond formed between two molecules is called intramolecular hydrogen bond.
Ø Hydrogen bond formed between two molecules is called intramolecular hydrogen bond.
Ø The bond dissociation energy of hydrogen bond in liquid water is about 23 kJ/mol.
Ø The bond dissociation energy of covalent bond between O and H in water molecule is 470 kJ/mol.
Ø Hydrogen bonds in water are very unstable and they frequently break and reform in liquid water.
Ø When one hydrogen bond breaks, another hydrogen bond is formed quickly with the same partner or another partner.
| You may also like... | ||
|---|---|---|
| NOTES | QUESTION BANK | COMPETITIVE EXAMS. |
| PPTs | UNIVERSITY EXAMS | DIFFERENCE BETWEEN.. |
| MCQs | PLUS ONE BIOLOGY | NEWS & JOBS |
| MOCK TESTS | PLUS TWO BIOLOGY | PRACTICAL |
Ø The life span of hydrogen bond in water is about 1 – 20 picoseconds (1 ps =10-12 s)
Ø Although individually weak, many hydrogen bonds collectively are responsible for the high cohesive force in water.
Ø The hydrogen bonds in water molecule are responsible for the UNUSUAL Properties of water.
Ø Intermolecular hydrogen bonds are responsible for the high boiling point of water (100oC).
Ø Intermolecular hydrogen bonds are responsible for secondary and tertiary structure of proteins and nucleic acids.
