Henderson-Hasselbalch equation is a simple expression which relates the pH, pKa and the buffer action of a weak acid and its conjugate base. The Henderson-Hasselbalch equation also describes the characteristic shape of the titration curve of any weak acid such as acetic acid, phosphoric acid, or any amino acid. The titration curve of a weak acid helps to determine the buffering pH which is exhibited around the pKa of that acid.
For example, in the case of acetate buffer, the pKa is 4.76. This is the best buffering pH of acetic acid. Besides, at this pH the acetic acid (CH3COOH) and acetate ions (CH3COO¯) will be at equimolar concentration in the solution. This equimolar solution of a weak acid and its conjugate base will resist the change in pH by donating or taking up the H⁺ ions. (pH is the negative logarithm of hydrogen ion concentration in a medium.The pKa is the negative logarithm of Ka. The Ka is the dissociation constant (similar to the equilibrium constant) for the ionization reaction of an acid.)
Learn more: Titration Curve of a Weak Acid (Acetic Acid)
In the present post, we will see the derivation of Henderson-Hasselbalch equation from the ionization reaction of a weak acid. We also discuss the significance of Henderson-Hasselbalch equation.
You may also like NOTES in...
BOTANY BIOCHEMISTRY MOL. BIOLOGY
ZOOLOGY MICROBIOLOGY BIOSTATISTICS
ECOLOGY IMMUNOLOGY BIOTECHNOLOGY
GENETICS EMBRYOLOGY PHYSIOLOGY
EVOLUTION BIOPHYSICS BIOINFORMATICS
Deriving Henderson-Hasselbalch Equation
Take the ionization reaction of a weak acid (HA):
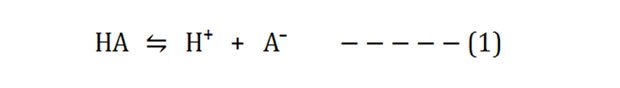
The dissociation constant Ka of the above reaction will be:
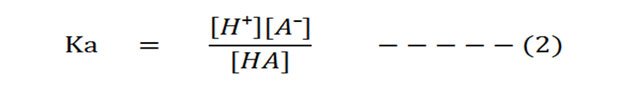
Dissociation constant is the ratio of the concentration of products by the concentration of reactants. The square brackets, [ ], denote ‘concentration’.
Then from the equation (2) take out the [H⁺] to the left side (solve for H⁺):
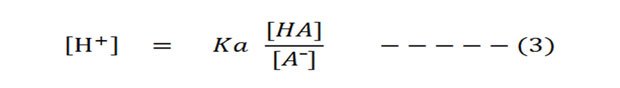 Then take the negative logarithm of both sides:
Then take the negative logarithm of both sides:
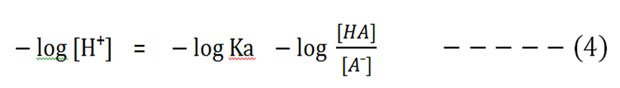
We know that the – log [H⁺] is pH and the – log Ka is pKa.
Substitute the pH and pKa in the equation (4):
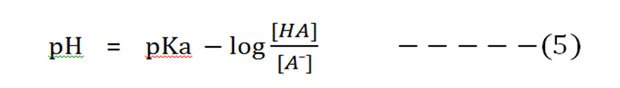
Now invert the -log [HA]/[A¯], which involves changing its sign to obtain the Henderson-Hasselbalch equation.
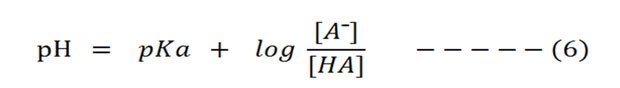
[A¯] is the concentration of proton acceptor (it can accept the proton in a buffer)
[HA] is the concentration of proton donor (it can donate a proton in the buffer)
Thus the equation (6) can be better stated as:
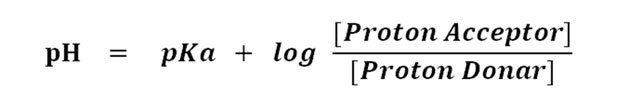
The above equation is called the Henderson-Hasselbalch equation.
Uses of Henderson-Hasselbalch Equation
The Henderson–Hasselbalch is mainly used for calculating the pH or pKa of a solution containing known quantities of a weak acid and its conjugate base.
Learn more: Biochemistry Solved Problems – pH, pKA and Henderson-Hasselbalch Equation.
| You may also like... | ||
|---|---|---|
| NOTES | QUESTION BANK | COMPETITIVE EXAMS. |
| PPTs | UNIVERSITY EXAMS | DIFFERENCE BETWEEN.. |
| MCQs | PLUS ONE BIOLOGY | NEWS & JOBS |
| MOCK TESTS | PLUS TWO BIOLOGY | PRACTICAL |
At the midpoint of the titration curve of a weak acid, the concentration HA (proton donor) equals the concentration of A¯ (proton acceptor). We have already stated that for a buffer the best buffering pH is at its pKa. (Also remember, the buffer is a mixture of equimolar concentration of weak acid and its conjugate base).
Let’s put it in the Henderson-Hasselbalch equation
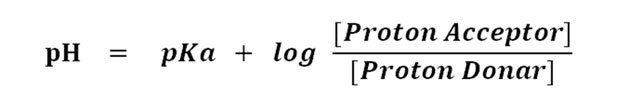
Since the concentration of proton acceptor (A¯) and proton donor (HA) is uniform in the midpoint of titration, the value becomes log 1.
Thus the equation becomes pH = pKa + log 1
log 1 = 0
Thus pH = pKa + 0
= pH = pKa
This also proved that for a buffer, the best buffering activity is obtained at the pH value equal to its pKa value.
References: Lehninger A.B., (2018), Textbook of Biochemistry, Ed. 5, Pearson International, New York
You might also like…
@. Titration Curve of Weak Acid
@. Workout Problems of pH and pKa using Henderson Hasselbalch Equation
@. Physical and Biological Properties of Water
@. How Hydrogen Bond is formed in Water?
