MCQ on Significance of Hydrogen Bonds. Hydrogen bonds are crucial in various aspects of chemistry and biology. They stabilize protein and DNA structures, facilitating life’s essential processes. In water, they contribute to its unique properties. In molecules like DNA, they enable genetic information storage and transfer, underpinning life’s molecular machinery. […]
Continue ReadingCategory Archives: Water
Proton Hopping in Water (Grotthuss Mechanism)
Proton Hopping in water is the process of diffusion of protons (H⁺ ions) through the network of hydrogen-bonded water molecules in the liquid water. Proton hopping is also called as Grotthuss mechanism, named after the discoverer Theodor Grotthuss. The net result of proton hopping is the fast movement of H⁺ […]
Continue ReadingpH and pKa – Henderson-Hasselbalch Equation Deriving
Henderson-Hasselbalch equation is a simple expression which relates the pH, pKa and the buffer action of a weak acid and its conjugate base. The Henderson-Hasselbalch equation also describes the characteristic shape of the titration curve of any weak acid such as acetic acid, phosphoric acid, or any amino acid. The […]
Continue ReadingPhysical and Chemical Properties of Water and its Biological Significance
Physical and Chemical Properties of Water: Water is the most abundant substance in the living system. Water makes up about 70% or more of the weight of almost all organisms. The life has originated in remote past in the aqueous environment. The properties (both physical and chemical) of water enabled […]
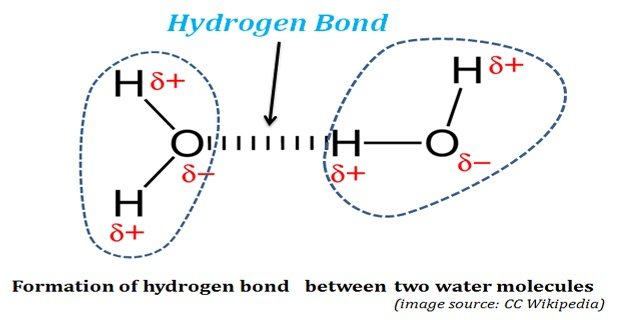
Continue ReadingHow Hydrogen Bond is Formed in Water?
Hydrogen Bonds in Water: The life was originated and started its evolution in water. Without water, life could not have existed on this planet. The properties of water, both physical and chemical, enabled water as the ‘solvent of life’. The water possesses some unusual physical and chemical properties. These ‘unusual […]
Continue Reading