Flowering plants (Angiosperms) show sexual reproduction. Flowers are the important reproductive structure produced by Angiosperms to affect sexual reproduction. Flowers are the objects of aesthetic and ornamental value, but for a botanist, flowers are the sites of sexual reproduction. This post is the Part-1 of Plus Two Botany Notes Sexual […]
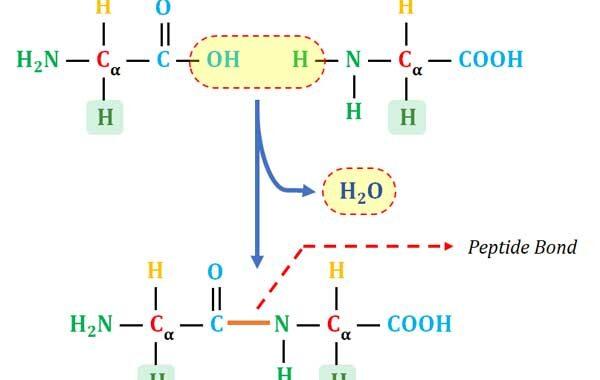
Continue ReadingFormation and Characteristics of Peptide Bond in Proteins
Learning Objectives: What are Proteins? How Proteins are Formed? What is Peptide Bond? Peptide vs Protein, C-Terminal and N-Terminal of Proteins, pI of Proteins, Sub-units of Proteins and Characteristics of Peptide Bond What are Proteins? Ø Proteins are the polymers of amino acids (specifically the polymers of L-α- amino acids). […]
Continue ReadingAchievements of Plant Breeding
In the previous post, we discussed the Objectives of Plant Breeding. Where we discussed the importance of plant breeding in providing food security and higher economic benefits. The present-day crops are very different from their wild relatives. This change in phenotype, higher yields, adaptability and suitability is brought about by […]
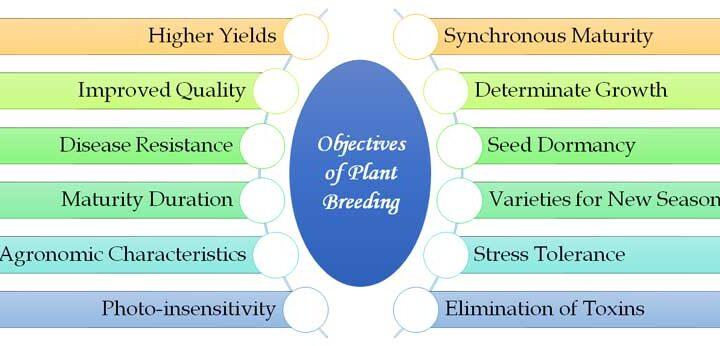
Continue ReadingObjectives of Plant Breeding
Plant breeding is the process of intentionally manipulating plant genetics to create new and improved plant varieties with desired traits. This involves selecting and crossing plants with desirable characteristics, such as higher yields, resistance to pests and diseases, improved nutritional content, or adaptation to specific environmental conditions. The present post […]
Continue ReadingWhat is Plant Introduction?
Plant introduction is the process of bringing plant species or varieties from one geographical location or ecological zone to another for the purpose of studying, evaluating, and potentially using them in agriculture, horticulture, or other industries. Plant introduction has been an important tool for plant breeding and crop improvement. It […]
Continue Reading