GATE XL Model Questions GATE XL Previous Year Papers
GATE Life Sciences (XL) 2010
(Graduate Aptitude Test in Engineering: Original Question Paper)
Biochemistry (I) Optional Paper
Questions 1 – 10 carries 1 mark each:
1. Nucleolus is involved in the synthesis of:
a. rRNA
b. tRNA
c. DNA
d. mRNA
2. In tryptophan operon, tryptophan acts as:
a. Repressor
b. Activator
c. Co-repressor
d. Co-activator
3. Positive selection of T cells ensures:
a. MHC restriction
b. Self-tolerance
c. TCR engagements
d. Activation by co-stimulatory signals
4. A DNA binding motif is:
a. Helix-loop-helix
b. Helix-turn-helix
c. Helical wheel
d. Loop-helix-loop
5. Amino acids responsible for N-linked and O-linked glycosylation of proteins are:
a. Asparagine and Aspartic acid
b. Glutamine and Serine
c. Glutamic acid and Serine
d. Asparagine and Threonine
6. One of the following compounds is NOT a neurotransmitter:
a. Dopamine
b. Glutamic acid
c. Histidine
d. Glycine
7. Approximate molecular weight (kDa) of the product after translation of a 390 base mRNA will be:
a. 48
b. 26
c. 39
d. 14
8. Lineweaver-Burk plot is a plot of :
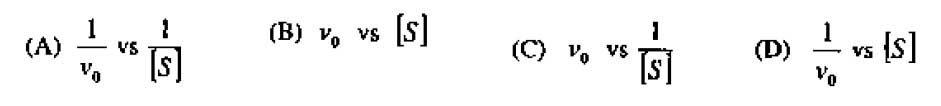
9. A mixture of proteins (W, X, Y, Z) elute form Sephadex G-200 column in the order W, X, Y, Z. The protein with maximum electrophoretic mobility on SDS-PAGE will be
a. W
b. X
c. Y
d. Z
10. Specific precursor of all prostaglandins is:
a. Oleic acid
b. Arachidonic acid
c. Palmitic acid
d. Α-linolenic acid
Questions 11 – 20 carry two marks each
11. Chymotrypsin and lysozyme are involved respectively in:
P. Removal of successive carboxyl terminal residues
Q. Hydrolytic cleavage of peptide bond
R. Cleavage of glycosidic C-O bond
S. Oxygen transport in blood
a. P, Q
b. Q, R
c. Q, S
d. R, S
12. Match the items in Group I with those in Group II
Group I Group II
P. Isotype switching 1. VH domain
Q. Clonal anergy 2. Non-responsive to self-antigen
R. Class II MHC 3. Non-responsive TH cells
S. Self-tolerance 4. β2-microglobulin
a. P – 1, Q – 4, R – 3, S – 2
b. P – 2, Q – 4, R – 1, S – 3
c. P – 1, Q – 3, R – 4, S – 2
d. P – 2, Q – 1, R – 3, S – 4
13. Multiple RNA polymerase transcribes a DNA template, unwinding about 1.5 turns of DNA template per transcription bubble. From the structural information of classical B-DNA, how many transcription bubbles are possible for a 180 base pair DNA molecule?
a. 12
b. 27
c. 6
d. 270
14. Match the items in Group I with most appropriate separation technique in Group II
Group I Group II
P. Mixture of glycine and albumin 1. Gas chromatography
Q. Mixture of 20 and 60 kDa proteins 2. Dialysis
R. Histones from nuclear extract 3. Affinity chromatography
S. Lectins 4. Size exclusion chromatography
5. Thin layer chromatography
6. Cation exchange chromatography
a. P – 1, Q – 4, R – 3, S – 5
b. P – 5, Q – 3, R – 6, S – 1
c. P – 2, Q – 4, R – 6, S – 3
d. P – 6, Q – 5, R – 2, S – 4
15. In the two half reactions:
Acetaldehyde + 2 H+ + 2e- → Ethanol ∆Eo = -0.16 V
NAD+H+ → NAD+ + 2H++ 2e- ∆Eo = -0.32 V
(F = 23,063 cal/V)
The ∆Go for the coupled reaction will be:
a. + 7400 cal
b. -7400 cal
c. -22200 cal
d. +22200 cal
16. Match the parameters in Group I with the correct option in Group II
Group I Group II
P. Km 1. Catalytic efficiency of the enzyme
Q. Kcat/Km 2. Affinity of enzyme to the inhibitor
R. pKa 3. Affinity of enzyme to the substrate
S. K1 4. Maximum buffering capacity
a. P – 3, Q – 1, R – 2, S- 4
b. P – 3, Q – 1, R – 4, S- 2
c. P – 1, Q – 2, R – 4, S- 3
d. P – 1, Q – 4, R – 2, S- 3
17. The rise per residue of α-helix is about 1.5 Å. A protein spans 2 nm bilayer, 7 times through its trans-membrane α-helical domain. Approximately, how many amino acid residues constitute the trans-membrane domain of the protein.
a. 105
b. 450
c. 30
d. 190
18. Match the protein in Group I with their correct function in Group II
Group I Group II
P. Shaker protein 1. Inner membrane receptor
Q. Bacteriorhodopsin 2. Active transport
R. Porin 3. Voltage gated K+ channel
S. ABC transporter 4. Light driven H+ pump
5. Membrane fusion
6. β- barrel simple diffusion channel
a. P – 4, Q – 2, R – 3, S – 5
b. P – 5, Q – 3, R – 4, S – 6
c. P – 6, Q – 1, R – 5, S – 4
d. P – 3, Q – 4, R – 6, S – 2
19. The metabolic disorders, alkaptonuria and phenylketonuria are caused by defects in the enzymes:
P. Glucose-6-phosphate
Q. Phenylalanine hydroxylase
R. Homogentisate 1, 2-dioxygenase
S. Tyrosinase
a. Q, R
b. P, R
c. P, Q
d. Q, S
20. Match the metabolic pathways in Group I with corresponding enzymes in Group II
Group I Group II
P. β-oxidation 1. Ribulose bisphosphate carboxylase
Q. Glyclysis 2. Phosphofructokinase 1
R. Gluconeogenesis 3. Phosphoenol pyruvate carboxykinase
S. Calvin cycle 4. Thiolase
5. Phosphofructokinase 2
a. P – 4, Q – 2, R – 3, S – 5
b. P – 3, Q – 2, R – 4, S – 1
c. P – 3, Q – 1, R – 5, S – 2
d. P – 4, Q – 2, R – 3, S – 1
Download all Previous GATE XL – Biochemistry Question Papers
We provide absolutely free online Study Materials for GATE XL (Life Sciences) examination to support the enthusiastic students to qualify the exam. You can use our GATE XL study materials like Books to Refer, Lecture Notes, Video Tutorials, PPTs, Model Questions (MCQ), Previous Year Questions, Mock Tests etc. for your preparation. Please remember, we can only provide the material stuffs and resources, the ultimate success depends on your dedicated preparation for the GATE exam. Feel free to ask any doubts/clarifications.
Free Lecture Notes and Study Materials for GATE XL Preparation


