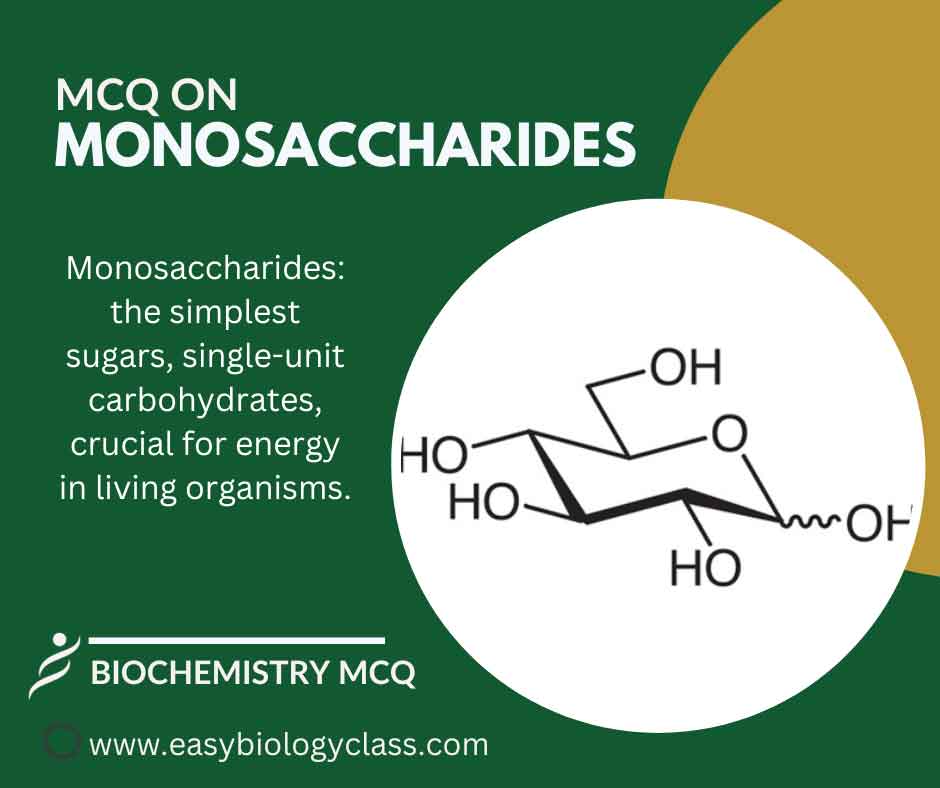
Monosaccharides are the simplest form of carbohydrates, consisting of single sugar molecules like glucose, fructose, and galactose. They serve as the basic units for more complex sugars and provide essential energy in biological processes. This is an MCQ on Monosaccharides Structure, Functions and Examples. Biochemistry Notes | Biochemistry PPTs | […]
Continue ReadingTag Archives: Monosaccharides
Monosaccharides: Definition, Structure, Characteristics, Classification, Examples and Functions
Monosaccharides are Simplest Sugars Monosaccharides are the simplest carbohydrates. They are polyhydroxy aldehydes or ketones with a carbon backbone. The carbon backbone in monosaccharides usually consists of 3 – 6 carbon atoms. The simplest monosaccharides are glyceraldehyde and dihydroxyacetone (with 3 carbons). The most abundant monosaccharide in nature is a […]
Continue Reading