Two Types of Apoptosis
In the previous post, we have discussed the characteristic features and significance of programmed cell death or apoptosis. As we discussed, the stimuli for the execution of programmed cell death can be of internal or external to the apoptotic cell.
Based on the source of stimuli, there are two types of apoptosis signaling pathways operate in the cells. They are (1) Intrinsic pathway (stimuli are internal) and (2) Extrinsic pathway (stimuli are external) of apoptosis. Even though both the intrinsic and extrinsic pathways considerably different, there is always cross-talk between these two pathways. In the present post, we will discuss the details of INTRINSIC PATHWAY of apoptosis signalling.
What is meant by Intrinsic Pathway of Apoptosis?
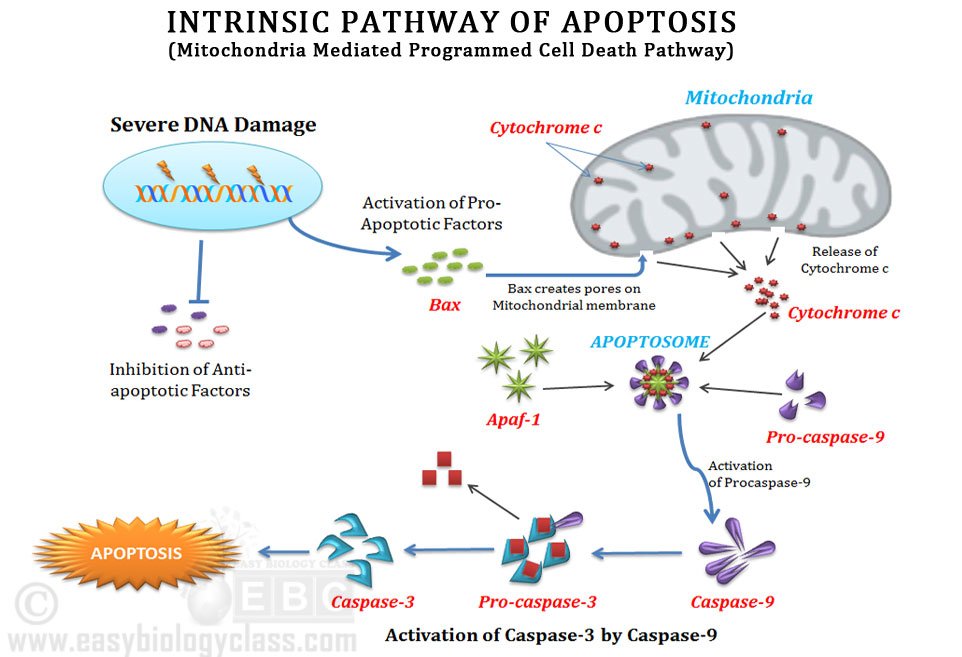
In the intrinsic pathway of apoptosis, the death-inducing stimuli are originated inside the target cell itself. Mitochondria, the powerhouse of the cell, have a significant role in executing the intrinsic pathway of apoptosis. Thus, the intrinsic pathway of apoptosis is also known as the Mitochondria-mediated death pathway.
| You may also like NOTES in... | ||
|---|---|---|
| BOTANY | BIOCHEMISTRY | MOL. BIOLOGY |
| ZOOLOGY | MICROBIOLOGY | BIOSTATISTICS |
| ECOLOGY | IMMUNOLOGY | BIOTECHNOLOGY |
| GENETICS | EMBRYOLOGY | PHYSIOLOGY |
| EVOLUTION | BIOPHYSICS | BIOINFORMATICS |
What are the stimuli for the intrinsic pathway of apoptosis?
Most commonly observed internal stimuli for the initiation of the intrinsic pathway of apoptosis are:
Ø Severe genetic damage
Ø Lack of oxygen (hypoxia)
Ø Very high concentration of cytosolic Ca2+ ions
Ø Presence of some viral proteins
Ø Severe oxidative stress due to the production of free radicals
What are Bcl-2 (B-cell lymphoma-2) family proteins?
The intrinsic pathway of apoptosis is facilitated by the members of Bcl-2 family proteins. The members of the Bcl-2 family proteins are characterized by the presence of one or more BH domains (Bcl-2 Homology Domain). The first identified member of Bcl-2 family proteins is Bcl-2 itself. The Bcl-2 was first identified as a cancer-causing oncogene in some human lymphomas. The gene which codes for the Bcl-2 protein was over-expressed in these cancer cells due to translocation. However, later studies have shown that Bcl-2 is not directly acting as an oncogene. They act as the oncogene by promoting the survival of the cancerous cells that would otherwise die by apoptosis.
The Bcl-2 family proteins were classified into THREE subcategories:
(1). Pro-apoptotic Bcl-2 proteins
(2). Anti-apoptotic Bcl-2 proteins
(3). BH3-only proteins
(1). Pro-apoptotic Bcl-2 members
Ø Pro-apoptotic Bcl-2 proteins promote apoptosis.
Ø In a normal cell, the pro-apoptotic Bcl-2 members are in inactive stage.
Ø Example: Bax and Bak
(2). Anti-apoptotic Bcl-2 members
Ø Anti-apoptotic Bcl-2 proteins inhibit apoptosis and ensure cell survival.
Ø Example: Bcl-2, Bcl-xL and Bcl-w
(3). BH3-only proteins
Ø BH3-only proteins only have a small BH3 domain.
Ø They can promote or inhibit apoptosis through an indict mechanism.
Ø Examples: Bid, Bad and Bim
The BH3-only proteins can be pro-apoptotic and they can promote apoptosis in two different ways. In some cases, they promote apoptosis by inhibiting the anti-apoptotic Bcl-2 members. In other cases, they stimulate apoptosis by activating pro-apoptotic Bcl-2 members. In both the cases, the BH3-only proteins are the key determinant in the cell survival or apoptotic cell death.
In a normal and healthy cell, the level of BH3-only proteins will be maintained in a very low concentration. Besides, in a healthy cell, the anti-apoptotic Bcl-2 proteins are able to restrain the pro-apoptotic members. If an apoptosis inducing stimulus is evoked in the cell, the levels of BH-3 only proteins are dramatically increased. This increase in the level BH3-only protein causes an imbalance in the level of pro-apoptotic and anti-apoptotic factors and beside that they shift the balance towards apoptotic cell death. When this balance is lost, the inhibitory effects of the anti-apoptotic Bcl-2 proteins are also compromised. In this critical situation, a mitochondria-mediate signaling cascade is initiated inside the cell and that will eventually result in the execution of programmed cell. The detailed mechanism of the intrinsic pathway of apoptosis is summarized below:
Ø When the cell lost the balance between pro-apoptotic and anti-apoptotic factors, a pro-apoptotic factor called Bax is translocated from the cytosol to the outer mitochondrial membrane.
Ø Bax protein undergoes a conformational change and gets inserted into the outer mitochondrial membrane.
Ø The assembly of Bax protein to the outer mitochondrial membrane is in such a way that it creates protein-lined channels or pores on the outer membrane.
Ø Due to the formation these pores, the permeability of the outer mitochondrial membrane dramatically increased.
Ø Through these pores, cytochrome c is released out of the mitochondria to the cytosol.
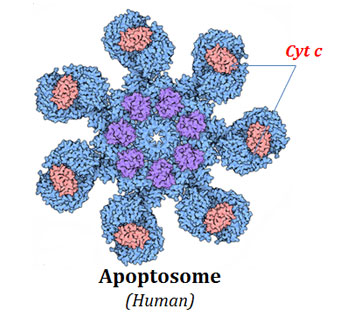
image source cc wikipedia
Ø The increase in permeability of the membrane also causes a dramatic loss in the electrical potential of the mitochondria.
Ø The loss of mitochondrial membrane permeability is accelerated by the increased level of Ca2+ ions in the cytoplasm which are released by the endoplasmic reticulum.
Ø The membrane potential compromised mitochondria also release SMACs (second mitochondria-derived activator of caspases) into the cytosol.
Ø SMACs bind to and inactivate all anti-apoptotic proteins in the cytosol.
Ø The complete release of cytochrome c to the cytoplasm is a ‘point of no return’. This means that the cell cannot be reverted back to its normal stage and moreover the cell should commit the apoptotic cell death.
Ø In the cytoplasm, the cytochrome c molecules combine together with Apaf-1 and Pro-caspase-9 in an ATP-dependent manner to form a multi-subunit complex.
Ø This multi-subunit complex of cytochrome c, Apaf-1 and pro-caspase-9 is called Apoptosome
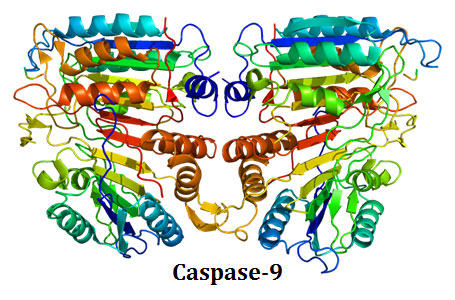
image source cc wikipedia
Ø A single apoptosome may contain several molecules of pro-caspase-9.
Ø The binding of Apaf-1 induce a conformational change in the pro-caspase-9 and it is activated to its fully proteolytic form (caspase-9).
| You may also like... | ||
|---|---|---|
| NOTES | QUESTION BANK | COMPETITIVE EXAMS. |
| PPTs | UNIVERSITY EXAMS | DIFFERENCE BETWEEN.. |
| MCQs | PLUS ONE BIOLOGY | NEWS & JOBS |
| MOCK TESTS | PLUS TWO BIOLOGY | PRACTICAL |
Ø Caspase 9 is the initiator caspases in the intrinsic pathway of apoptosis.
Ø Caspase 9 activates the executioner caspases such as caspases -3 and Caspase-7.
Ø Activated caspase-3 and caspase-7 cleaves its target molecules in the cell and thus the apoptotic cell death is executed.
<<Back to Molecular Biology Notes

It is a very good explanation and presentation with diagram of intrinsic pathway. I felt hard for understanding it by reading in the book even after reading it pin to pin. But after reading this I got a good and clear idea . Thank you
i felt hard studying these pathways in my book. so i thought of searching it in Internet. luckily i have got this easy biology class and i read the pathways in this. It was really easy for me to study and take notes from this site. I have studied the pathways within 30 mins time span.
Thank you Fathima, Keep visit easybiologyclass