
.

source: The Hindu
Artemisinin is an anti-malarial parasite drug extracted from a Chinese medicinal plant called sweet wormwood (Artemisia annua; Asteraceae family). It is also known as qinghaosu in Chinese and it is the most rapid acting drug currently available against the malarial parasite, Plasmodium falciparum.
The 2015 Nobel Prize in Physiology and Medicine is shared by William C. Campbell, Satoshi Omura and Youyou Tu for the discoveries of therapies against parasitic infections which saved millions of life over the globe. Among the three Nobel laureates, Professor Youyou Tu of China shares half of the Prize money for the credit of isolation, purification and structure elucidation of Artemisinin, the anti-malarial parasite drug for the first time from a Chinese traditional medicinal plant Artemisia annua. Professor Tu is the first China based Woman scientist to win the science Nobel.

source: wikipedia
Discovery
Artemisia annua (sweet wormwood) is a herbal plant found in many parts of the world. The therapeutic potential of this plant has been known to Chinese people for at least 2000 years. It has been widely employed in the Chinese Traditional system of medicine against a number of infectious diseases. The scientific discovery of Artemisinin in 1972 by Professor Youyou Tu was done as a part of a plant screening research program launched in 1967 under the name ‘Project 523’ set by the Chinese Government. ‘Project 523’ is a code name of a secret military project by Chinese government during Vietnam war for finding a cure against malarial parasite as per the directs from Mao-Zedong. A sub-division of the project team headed by Professor Youyou Tu selected and screened more than 2000 plants for its antimalarial properties based on references in Chinese Traditional Medicine. Tu’s group discovered that the ethanol extract from Artemisia annua was promising and the extract was found to inhibit rodent malarial parasites. The efficiency of the first extracted crude extract of Artemisia was very low (12 – 40%). So again Professor Tu turned to ancient Chinese literature and studied the recipes of Ge Hang, 340 AD. From there she got a recipe for the cold extraction of the plant using cold water. With this she developed another efficient low temperature extraction method with ether. With the new cold extract, Professor Tu was able to achieve almost 100% killing of malarial parasite in in vivo condition in mice. Latter the crude extract was purified to isolate the active ingredient and named it as Artemisinin. The ‘Project 523” also developed other drugs that are used in combination with Artemisinin to treat malaria include, lumefantrine, piperaquine and pyronaridine. Their results were published in Chinese Journal of Medicine in 1976. In the late 1990s, Novartis (a multinational pharmacological company) had patented the first Artemisinin based combination therapy (ACT) along with artemether and lumefantrine.
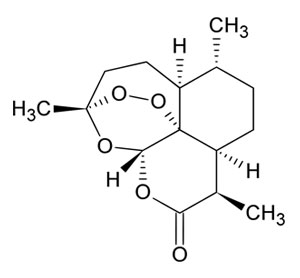
Chemistry of artemisinin:
source: wikipedia
Chemically artemisinin (C15H22O5) is a sesquiterpene lactone containing an unusual peroxide bridge. The peroxide bridge is known to be responsible for the antimalarial properties of Artemisinin. A very few natural compounds are known to contain the peroxide bridge.
Mechanism of action of artemisinin:
Artemisinin is a prodrug, it is metabolically converted into a bioactive form called dihydroartemisinin (commonly called as arteminol) and it is responsible for the killing of malarial parasite at the stage when the parasite is located inside the red blood cells of the host. The exact mechanism of action of dihydroartemisinin is not known. There are many possible mechanisms and many of which are only theoretical predictions without any strong experimental supports.
Most of the experimental evidences support the free radical mechanism of the peroxide bridge in the artemisinin. Although highly criticizing, a possible mechanism of action describes that, during the pathogenesis of malarial parasite, the pathogen consumes hemoglobin. In the digestive vacuole of the parasite, the iron group of hem reduces the peroxide bond of artemisinin. This generates high-valent-oxo reactive species, which triggers a series of reactions to produce more reactive oxygen radicals which ultimately kill the pathogen. This proposal is highly criticized in the scientific community; since it is not referring the specificity of the reaction and more than that the free radicals generated are highly toxic to the host cells also.
There other possible mechanisms of action of artemisinin are:
Ø By disturbing the cellular redox cycling by inhibiting Exported Protein 1 (EXP1), a membrane glutathione-s-transferase.
Ø By inhibiting a calcium-ATPase gene of Plasmodium falciparum (PfSERCA)
Ø By inhibition of cysteine proteases in the digestive vacuole of the parasite
Ø By accumulating hemoglobin in the parasite and inhibition of hemozoin formation
Ø By the damage of the vacuolar membrane of the parasite
Ø By disturbing the mitochondrial electron transport chain of the parasite
Even though the exact mechanism of action of artemisinin is not known to science, the ACT treatment dramatically reduce the mortality rate of malaria by 47% and also the number of malarial infection dropped to 26% from 173 million to 128 million (WHO, 2014). Today ACT treatment reduced the incidence of malaria among millions of people worldwide. Due to the poor bio-availability of artemisinine in the cells, nowadays a combination therapy called ACT (artemisinin-combination therapies) is preferable against malarial infections.
Artemisinin, a possible anti-cancer drug:
A probable emergence of artemisinin in the near future may be by its ability to cure cancer. The probable mechanism of action is by the release of free radicle. When artemisinin is reacted with the iron, it can release free radicals which can be used as a chemotherapeutic strategy to kill cancerous cells. Normally cancerous cells requires and uptake large amount of iron to proliferate than the normal cells. Thus they will be more susceptible to the cytotoxic effect of artemisinin than the normal cells. More than that cancerous cells also express a large number of transferrin receptors that facilitate the uptake of plasma containing iron carrying protein called transferrin by endocytosis (transferrin is an iron binding blood plasma glycoprotein that control the level of free iron in biological fluids). Researchers are now able to induce apoptosis in cancerous cells by treating the cells with transferrin which is covalently bounded to artemisinin. Thus Artemisinin and Artemisinin tagged iron carrying components could be developed into potential anti-cancerous agents in the near future.
Artemisinin: The Wonder Phyto-drug against Malarial Parasite Plasmodium falciparum.
The Nobel Prize winning contribution of Professor Youyou Tu, China in the 2015 Nobel Prize in Physiology and Medicine
Please Share for your Students, Colleagues, Friends and Relatives…
