Biochemistry MCQ-04: Amino acids are the building blocks of proteins. They are organic compounds that contain both an amino group (-NH2) and a carboxyl group (-COOH) attached to a central carbon atom. There are 22 different types of amino acids that are commonly found in proteins, each with a unique side chain that gives it distinct properties. In addition to their role in protein synthesis, amino acids also play important roles in metabolism, cell signaling, and other biological processes. This the Part-1 of the Multiple Choice Questions MCQ on Amino Acids with answer key and explanations
You May also like: Amino Acids MCQ Part-1 | Part-2 | Part-3 | Part-4 | Part-5 |
(1). Which of the following amino acid is sweet in taste?
a. Glycine
b. Alanine
c. Glutamic acid
d. None of these
(2). Which of the following amino acid act as inhibitory neurotransmitter?
a. Glycine
b. Alanine
c. GABA
d. All of these
e. None of these
(3). The amino acid commonly used as an ingredient in the buffers of SDS PAGE.
a. Aspartic acid
b. Glutamic acid
c. Glycine
d. Aspartic acid and Lysine together
(4). Which of the following amino acid will be absent in α (alpha) helix structure of protein?
a. Glycine
b. Galine
c. Glutamic acid
d. Proline
(5). Aminolevulinic acid, the first product in porphyrin biosynthesis in eukaryotes, is synthesized from ____ and succinyl-CoA.
a. Valine
b. Tryptophan
c. Methionine
d. Glycine
(6). Carnosine is a dipeptide of histidine and _____.
a. β-histidine
b. β-alanine
c. β-lysine
d. α-lysine
e. Tryptophan
(7). Which of the following enzyme contain Selenocysteine?
a. Nitrate reductase
b. Catalase
c. Glutathione peroxidase
d. All of these
(8). Amino acid used in the ‘stripping’ of Western blotting experiment is:
a. Glutamic acid
b. Phenyl alanine
c. Alanine
d. Glycine
(9). Total number of proteinogenic (protein building) amino acids in the living world is ____.
a. 20
b. 21
c. 22
d. 23
(10). Amino acid selenocysteine is coded by_______.
a. UAA
b. UAG
c. UGA
d. AUG
(11). Which of the following is a glucogenic amino acid?
a. Glycine
b. Proline
c. Alanine
d. Both (a) and (b)
e. All of these
(12). Which of the following amino acid act as excitatory neurotransmitter?
a. Glutamate
b. Aspartate
c. Cysteine
d. All of these
e. None of these
(13). Which amino acid act as the precursor of nucleotide biosynthesis?
a. Aspartate
b. Glycine
c. Glutamine
d. Both (a) and (b)
e. All of these
(14). Bacteria prefer to use the codon CGA instead of AGA to code for Arginine. This is an example for _____.
a. Second genetic code
b. Nullomers
c. Transcriptional decoding
d. Allomers
(15). Which of the following is an example for a bio-plastic?
a. Polyaspartate
b. Polyglutamate
c. Poly-L-lysine
d. All of these
(16). Histones are rich in__________.
a. Lysine
b. Arginine
c. Histidine
d. Lysine and Arginine
(17). Single letter code of pyrrolysine is _______.
a. B
b. J
c. O
d. U
You may also like NOTES in...
BOTANY BIOCHEMISTRY MOL. BIOLOGY
ZOOLOGY MICROBIOLOGY BIOSTATISTICS
ECOLOGY IMMUNOLOGY BIOTECHNOLOGY
GENETICS EMBRYOLOGY PHYSIOLOGY
EVOLUTION BIOPHYSICS BIOINFORMATICS
(18). Which of the following is not an essential amino acid?
a. Proline
b. Histidine
c. Leucine
d. Methionine
(19). What is the molecular weight of Glycine?
a. 75 g·mol−1
b. 80 g·mol−1
c. 90 g·mol−1
d. 95 g·mol−1
(20). Among the 20 standard proteins coding amino acids, which one is least occurs in proteins?
a. Glycine
b. Alanine
c. Tryptophan
d. Methionine
Answers with explanations
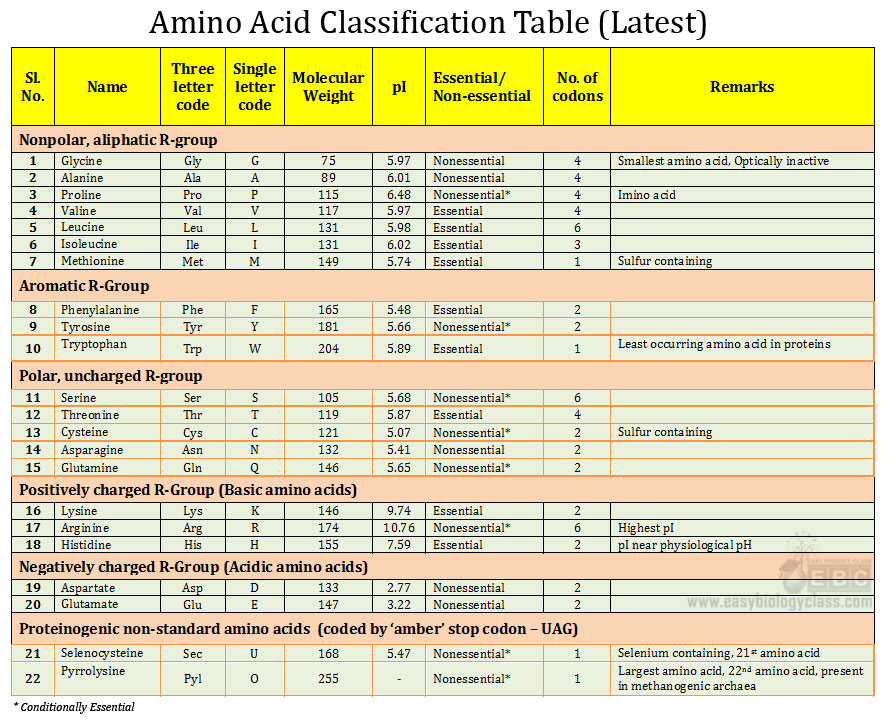
1. Ans. (a). Glycine
2. Ans. (d). All of these
Inhibitory amino acid neurotransmitters (IAA): GABA, Glycine, β-Alanine, Taurine
Excitatory amino acid neurotransmitters (EAA): L-Glutamate, L-Aspartate, L-Cysteine and L-Homocysteine
3. Ans. (c). Glycine
Depending up on the pH of buffer, glycine can exist in three different forms (positively charged, no charged and negatively charged)
4. Ans. (d). Proline
Proline is an imino acid. The amino group of proline is covalently connected to the side chain (R group) and thus it reduces the rotational flexibility. Furthermore proline produces a kink in the polypeptide. Due to these reasons proline is very rarely present in α-helixes.
5. Ans. (d). Glycine
6. Ans. (b). β-alanine
Carnosine, dipeptide of histidine and β-alanine, is an antioxidant present in muscle cells and brain cells of human.
β-alanine is naturally occurring β-amino acid (amino group is at the β position not in the α position)
7. Ans. (c). Glutathione peroxidase
Selenocysteine (Sec) is considered as the 21st protein coding amino acid. It is a rare amino acid containing Selenium as selenol group (SeH). Example of proteins containing selenocysteine: glutathione peroxidases, thioredoxin reductases, formate dehydrogenases, glycine reductases, selenophosphate synthetase and methionine-R-sulfoxide reductase.
Selenocysteine is coded by UGA codon, one of the stop codon, by a mechanism called translational recoding.
8. Ans. (d). Glycine
‘Stripping’ in western blotting is a procedure of removing protein bounded antibodies from the membrane so that the membrane can be reused to probe with another antibody and hence reduces the sample processing time.
9. Ans. (c). 22
There are a total of 22 protein coding amino acids, among these 20 are coded by standard genetic code (standard amino acids). The remaining two amino acids (Selenocysteine and Pyrrolysine) are coded by two stop codons.
Selenocysteine is considered as the 21st amino acid coded by UGA codon
Pyrrolysine is considered as the 22nd amino acid coded by UAG codon
10. Ans. (c). UGA
11. Ans. (e). All of these
Glucogenic amino acids are those amino acids which are converted into glucose by gluconeogenesis during fasting and starvation. Except Leucine and Lysine, all amino acids are glucogenic.
Ketogenic amino acid: Amino acids which cannot be converted into glucose during catabolism. These amino acids are directly converted into Acetyl CoA through ketogenesis and enters into Kreb’s cycle for energy release
Leucine and Lysine are exclusively ketogenic amino acids.
Some amino acids such as Isoleucine, Threonine, Phenylalanine, Tyrosine and Tryptophan are both glucogenic and ketogenic.
12. Ans. (d). All of these
13. Ans. (e). All of these
14. Ans. (b). Nullomers
Nullomer: They are codons of natural amino acids but are generally not present in the genome of the organism even though these codons are theoretically possible. For example, AGA and CGA are the codons for arginine in all organisms. However, in most bacterial species the codon AGA is not used for coding arginine and if we artificially substitute AGA for CGA, it will be lethal to the organism.
Second genetic code: is a general explanation for two biological events.
(1). How amino acid residues in a protein determines the secondary and tertiary conformation?
(2). The mechanism of tRNA specificity to its correct amino acyl tRNA synthase enzyme for the activation of amino acid during the initiation of translation.
Transcriptional decoding: A mechanism in the cells by which the stop codons are used for coding 21st (Selenocysteine) and 22nd (pyrolysine) amino acids during protein synthesis.
Allomeres (Allomerism): Formation of same type of crystalline structures by chemically different molecules.
15. Ans. (a). Polyaspartate
16. Ans. (d). Lysine and Arginine
Histones are proteins of eukaryotes which help in the packing of DNA in nucleosome. Histones are highly basic in charge since they are rich in basic (positively charged) amino acids such as lysine and arginine. Positive charges of histones stabilize the negatively charged DNA by charge-charge interactions. Chemical modification of histones (acetylation and methylation) helps in the winding or unwinding of DNA during transcription, replication and repair.
17. Ans. (c). O
Single letter code of selenocysteine is U
18. Ans. (a). Proline
Essential amino acids: Histidine, Isoleucine, Leucine, Lysine, Methionine, Phenylalanine, Threonine, Tryptophan, Valline
Non-essential amino acids: Alanine, Arginine, Asparagine, Aspartic acid, Cysteine, Glutamic acid, Glutamine, Glycine, Proline, Serine and Tyrosine
Conditionally essential amino acids (= essential in certain instances such as infants or during some diseases): Arginine, Cysteine, Glutamine, Pyrrolysine, Proline, Selenocysteine, Serine and Tyrosine
19. Ans. (a). 75 g•mol−1
Glycine is the smallest amino acid
Tryptophan is the largest amino acid among standard amino acids with molecular weight 204 g·mol−1
Molecular weight of pyrrolysine is 255 g.mol-1
20. Ans. (c). Tryptophan
You May also like: Amino Acids MCQ Part-1 | Part-2 | Part-3 | Part-4 | Part-5 |
You may also like...
NOTES QUESTION BANK COMPETITIVE EXAMS.
PPTs UNIVERSITY EXAMS DIFFERENCE BETWEEN..
MCQs PLUS ONE BIOLOGY NEWS & JOBS
MOCK TESTS PLUS TWO BIOLOGY PRACTICAL
